TOP 10 FACTORS TO CONSIDER FOR SELECTING
QMS Software
For Medical Device
and Pharma Companies.
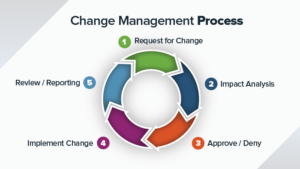
There are many challenges involved when selecting and implementing a Quality Management System (QMS) software for a medical device company. Even in a small to mid-size company there are multiple departments and employees with the different goals. You would need to document your user requirement specifications, at least at a higher level, so that you can communicate properly to the vendors. This will help you select a right size quality management system (QMS) software for your company. Here are the top 10 factors you should consider for the selection and implementation of a quality management system (QMS) software for medical device and pharma companies. If you are rolling out a QMS system to comply with ISO13485 standard, please refer to our blog post for understanding the top 10 requirements for ISO13485.


If you are running a Quality Management System with paper based or Excel spreadsheets, it may be taking lot of your valuable time for tracking and recording data for Quality Management system (QMS) activities. The manual systems can lead to compliance issues, sometimes people forget to complete the records, or simply misplace the records. Though this is challenging, still many companies manage the QMS with manual paper-based systems.
These activities become more challenging when you are managing a QMS for a medical device or pharma manufacturing company. A well-organized electronics Quality Management System (QMS) software can be a big help to automate the QMS processes and tasks. One thing to keep in mind that no one size fts all. There are many QMS software providers for medical device and pharma manufacturing applications. You have to pick the one that best meets your needs and requirements.
Manual QMS


- Paper based and error prone
- Excel or Access database
- No visibility of performance
- Audit issues
- Resource wastage
Qualcy eQMS


- Simple and easy to use
- Save time and money by 80% or more
- Access anytime, anywhere
- Built in compliance for FDA/EU Regulations
The following sections describes top 10 factors you should consider for the selection and implementation of a quality management system (QMS) software for medical device and pharma companies.




If you are using the quality management system (QMS) software for medical device and pharma manufacturing applications, the QMS software has to be validated and qualified prior to use. What level of validation is required? The validation should be done based on a risk analysis of your processes and workflows. Usually, the validation is done by the QMS software service providers. You must ensure that your use cases and workflows have been included in those validation reports.




There are mandatory requirements and guidelines available for the validation of a Quality Management System (QMS) software as per the ISO13485 standards and FDA regulations.
The validation reports should be available for the compliance audits. We have discussed the requirements requirements for the qualifications and validations of the QMS software in a separate article.





The quality management system (QMS) software shall comply with 21 CFR part 11 requirements. There are specific requirements under 21 CFR part 11, which governs use of electronics records and electronic signatures in the medical device and pharma applications. Electronics records shall follow the general guideline of the ALOCA rule. The acronym ALCOA stands for A: attributable, which means when a record is created by an individual, there shall be traceability of linking the record and the individual, L:Legible record, which means the electronics records shall be created in a format that is readable and retainable for long periods of time, i.e in a PDF or XML format, C: Contemporaneous, which means the electronics records had to be created at the time of the event, the electronics records shall not be created by transcribing or copying another record. O:Original, which means the electronics records shall be in the original form. When there is any changes made, those changes shall be available in the audit trails. A: accurate records, which means the electronics records will be kept inaccurate form. There will not be any option for modifying or changing the electronics records.
The electronic signatures are equivalent to legally binding wet signatures on the paper. We have covered the requirements for 21 CFR part 11 in a separate article.




One of the key requirements for quality management system (QMS) software is the usability and intuitiveness of the software interface. If it is hard to understand, or it is going to take too much efforts to understand, the users are not going to like it. Then you may have challenges convincing the users to use the software. What is the point of buying an expensive piece of QMS software if the users are not going to use it. So, you must ensure the QMS software has good usability features. The best way to do this, is to select a software and then do a dry run with some features and applications and see how it feels. This will give you good idea whether the software is well designed or not.
Customization and maintainability: When you select a piece of software, it is never going to be the perfect version of what you are looking for or what your users want. You would need some form of customizations, for example you may need to change the description of the labels, or you may need to change the dropdown list, so that it will match your current process. You should select a QMS software that fits to your requirements. You should never fit your requirements into the QMS software, because that is a wrong strategy. It will have long term challenges when managing the system, This will create more work rather than the software helping you simplifying your systems and saving your time.




Now-a-days most of the QMS software are being provided through the cloud services. You have to make sure your data is secured because you have ultimate responsibility for the security of your data. You have to ensure the software service provider has appropriate controls for the backup and security of your data.




When you select the QMS software for medical device application, you should get good training and supports from the service provider. This will help you to do smooth migration of your existing data. In case you have any questions, when using the QMS software, you should get the support from the service providers. This support should be readily available, the support staff should be knowledgeable about the functionalities of this software. The support staff should have good understanding of the included requirements. Most of the QMS software companies do not have qualified and trained people to provide support services.




When you are going to use the QMS software for the medical device and pharma applications, there are some key functionalities you must be looking for when selecting the QMS software. The key functionality’s include user management and access control, Design history file and Risk management, Document change control and Training record management, Corrective action and Preventative action (CAPA), Complaint management, Supplier Quality Management and others.




You must have option for controlling the access of the users. Only the authorized users should have access to making changes and approving the documents. you must control the access and permissions of the users, who are going to use the quality management system (QMS) software.




for the medical device applications you will be creating design input files, design output files, design review records, creating risk management files and others. Integrating all the design documents takes a lot of time and efforts. The QMS software should help you manage the design files and risk management activities in a seamless and integrated manner. We will cover the requirements for the design history files and risk management activities involved in a medical device application in a separate article.



The QMS software used in medical device should have functionality for document change controls, which should be integrated with training management. When you are making changes to the documents, you should have option for creating training records and sending the training assignments to the users as part of the document change control system. The training records will provide evidence that the users have been informed about the changes before the changes become effective. We’ll cover the details of the document change control and training requirements in medical device applications in a separate article.




The QMS software should have a module for managing complaint records. this is a key regulatory requirement that many medical device companies have failed to comply with. You should have the option at minimum, for recording the complaints by date, name of the complaint, type of complaint, decision to investigate the complaint, decision to recall the products or not to recall the products, details of the investigation completed, details of the corrective action taken, decision to report the incident two regulatory bodies.




When you are selecting any QMS software for medical device applications, The service provider should have good track record of previous roll out of the QMS software applications. This provides good evidence that the software has been qualified and validated. Here is a sample list of QMS Software providers as listed in the review sites.


In summary it is your responsibility to select a proper QMS software for your medical device or pharma manufacturing company that will meet your needs and requirements. The QMS software should also help you comply with regulatory requirements. If you need any help, you can check out our other articles and video postings. For more information about Qualcy QMS software, contact us for a demo/free trial.
Check out the YouTube Video to learn more about the QMS software for Medical Device


About the Author
Sanjay Dhal
Sanjay is a Quality Management Professional with more than 20 years’ experience in the Medical Device and other manufacturing industries. Currently Sanjay is serving as Group leader of Quality & Compliance Solutions at Qualcy Systems Inc. He has helped 100+ companies set up effective QMS and CMMS systems. Sanjay is a ASQ certified six sigma black belt (CSSBB), CQE, CQA, and has MS and MBA degrees.